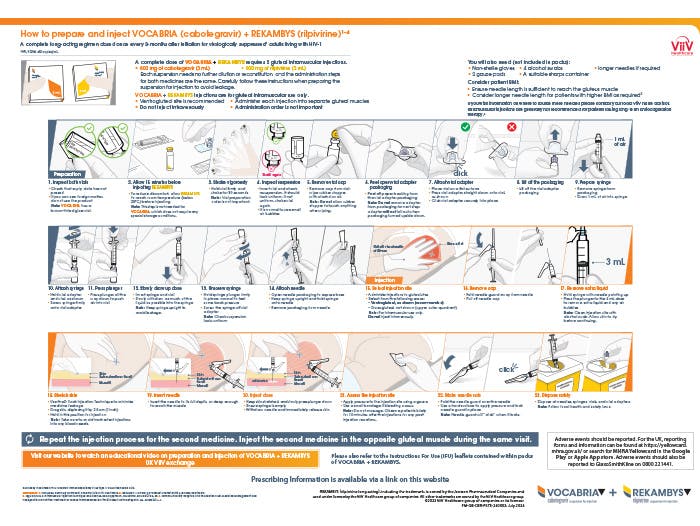
A range of resources to help successfully integrate 2-monthly VOCABRIA + REKAMBYS into your clinic
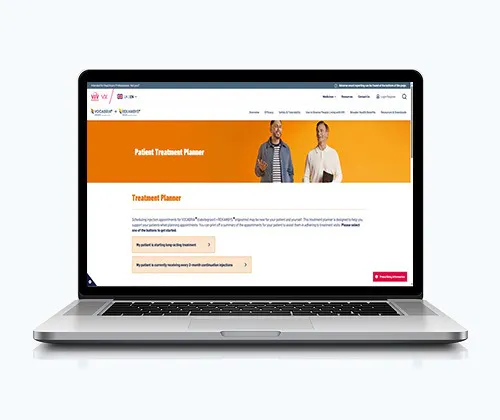
Treatment Planner
The Treatment Planner has been designed to make scheduling appointments for every-2-month VOCABRIA + REKAMBYS easier for you and your team.
Get started
Preparation and injection tools
A summary of preparation and injection steps that can be viewed online.
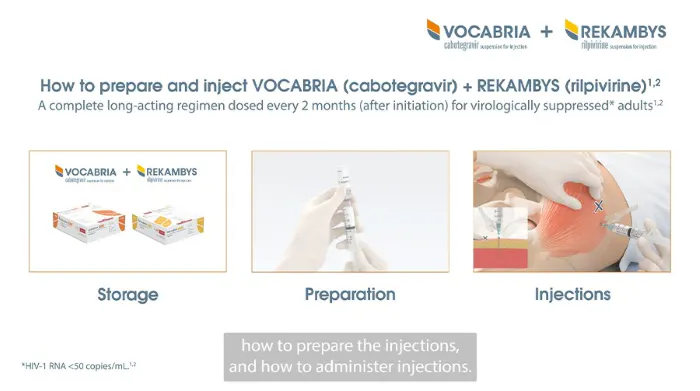
Administration Video
As the correct preparation and administration is important, we are here to help you. Watch this video to learn more.
To support you when deciding if a patient might be suitable for Vocabria and Rekambys.
To support you in the implementation of Long-acting Injectables (LAIs) into your clinical setting.
BIC/FTC/TAF=bictegravir/emtricitabine/tenofovir alafenamide; BMI=body mass index; FAQ=frequently asked question; HCP=healthcare professional; ID=identification
References:
- VOCABRIA (cabotegravir) 600 mg suspension for injection Summary of Product Characteristics (SmPC)
- REKAMBYS (rilpivirine) 900 mg suspension for injection Summary of Product Characteristics (SmPC)
- Kara D, Uzelli D, Karaman D. Using ventrogluteal site in intramuscular injections is a priority or an alternative? Int J Caring Sci. 2015;8(2):507–513.
REKAMBYS (rilpivirine long-acting injection), including the trademark, is owned by the Janssen Pharmaceutical Companies and used under license by the ViiV Healthcare group of companies. All other trademarks are owned by the ViiV Healthcare group.
PM-GB-CBR-WCNT-250011 v2 | March 2026
Adverse events should be reported. Reporting forms and information can be found at https://yellowcard.mhra.gov.uk/ or search for MHRA Yellowcard in the Google Play or Apple App store. Adverse events should also be reported to GSK via the GSK Reporting Tool or on 0800 221441.